Introduction & Approach
- Life threatening acute complications of MI includes the following differential:
- Mechanical Complications:
- LV free wall rupture –> Leads to cardiac tamponade
- Ventricular septal defect –> Leads to RV volume overload and shock
- Papillary muscle rupture –> Leads to acute severe mitral regurgitation, causing pulmonary edema and shock.
- LV pseudoaneurysm (aka “contained rupture”) – myocardial rupture, but saved by pericardial adhesions.
- Non-Mechanical Complications
- AV block –> Usually intermittent due to vagal etiology, but sometimes due to direct ischemia/infarction, which is permanent.
- RV Infarct –> Severe RV failure, classically presenting with hypotension/shock, RV volume overload and no pulmonary edema.
- LV thrombus –> Very high risk of stroke.
- Mechanical Complications:
- A mechanical complication of MI must be suspected if:
- The patient presents in shock or acute pulmonary edema
- A prominent murmur is auscultated
- Late presentation MI after resolution of ischemia
- If a mechanical complication is suspected:
- Urgent structural assessment is required with a bedside echocardiogram/POC-US.
- The benefit of a second antiplatelet agent must be reassessed. ASA is usually acceptable, however if a mechanical complication is highly suspected, a second antiplatelet agent may need to be withheld until further assessment with urgent cardiac imaging. If surgery is required, second antiplatelet agent should be avoided, if possible, to reduce the risk of operative bleeding.
- Clinicians may choose a short-acting anticoagulant, such as heparin, which can be easily reversed if urgent surgery is needed.
Left Ventricular (LV) Thrombus
- LV thrombus is usually seen in patients presenting with large anterior STEMIs
- Late presentation is an important risk factor
- LV thrombus results from the “perfect storm” of factors contributing to “Virchow’s Triad:”
- LV wall akinesis/dyskinesia resulting in stagnant blood flow
- Pro-coagulative state
- Ischemia resulting in inflammation
- The incidence of LV thrombus is about 5-7% in anterior MI.
- (Number may be smaller with modern reperfusion times)
- Imaging options:
- Echo (most commonly used)
- CT or MRI.
- LV thrombus forms 12-72 hours after MI.
- Imaging too early in the MI course may not identify an LV thrombus
Treatment of LV Thrombus
- The most dreaded complication of LV thrombus is stroke.
- Treatment of LV thrombus
- Vitamin K antagonists (VKAs)
- DOACs are available, but only used if VKA-intolerant (no clinical trials available, case reports only)
- Prevention of LV thrombus in anterior MI
- CCS 2018 – No routine anticoagulation (citing poor evidence and high risk of bleeding)
- AHA 2013 – Class IIB indication for anticoagulation
- Generally treat with anticoagulation for 3-6 months, and repeat echo to reassess whether the LV thrombus has resolved.
- Chronic LV thrombus (> 3-6mo) often becomes “mural”, and has a significantly lower embolic risk. Many clinicians switch to a DOAC at that point to reduce the risk of bleeding, however no clinical practice guidelines or clinical evidence helps guide the management of chronic LV thrombus (as of Jan 2021).
| CCS 2018 Heart Failure Guidelines |
|---|
|
| AHA 2013 STEMI Guidelines |
|---|
|
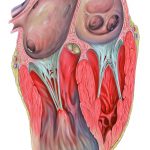
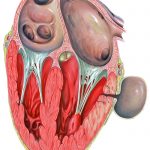
Left Ventricular Aneurysm and Pseudoaneurysm
- Left ventricular aneurysm
- Result of thinning of the myocardium
- Identified by a large neck
- Low risk of rupture
- Persistent ST elevation on EKG
- Left ventricular pseudoaneurysm
- Cardiac myocardial rupture that is contained by the pericardium
- Very narrow neck
- High risk for rupture → increased mortality
Right Ventricular (RV) Infarct
- Complication of inferior STEMIs
- Loss of RV contractility to maintain forward flow leads to LV underfilling (reduced preload), reduced cardiac output, and hypotension.
- Drugs that further reduce preload such as nitrates and diuretics can worsen cardiac output leading to worsening hypotension and shock.
- The RV is supplied mostly by the RV branch of the RCA. A proximal RCA lesion can compromise the RV branch.
- Early recognition and diagnosis of an RV infarcts is important.
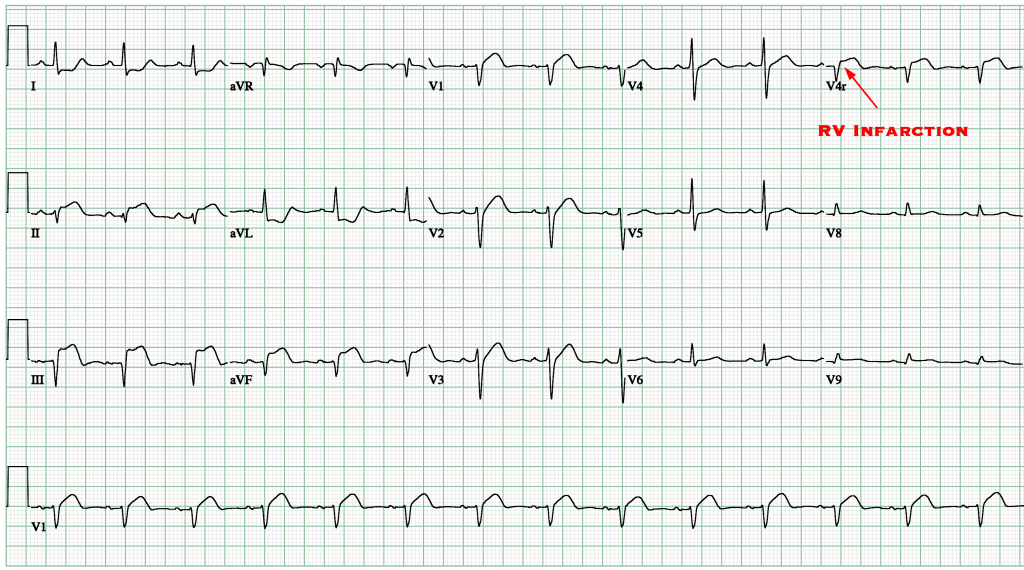
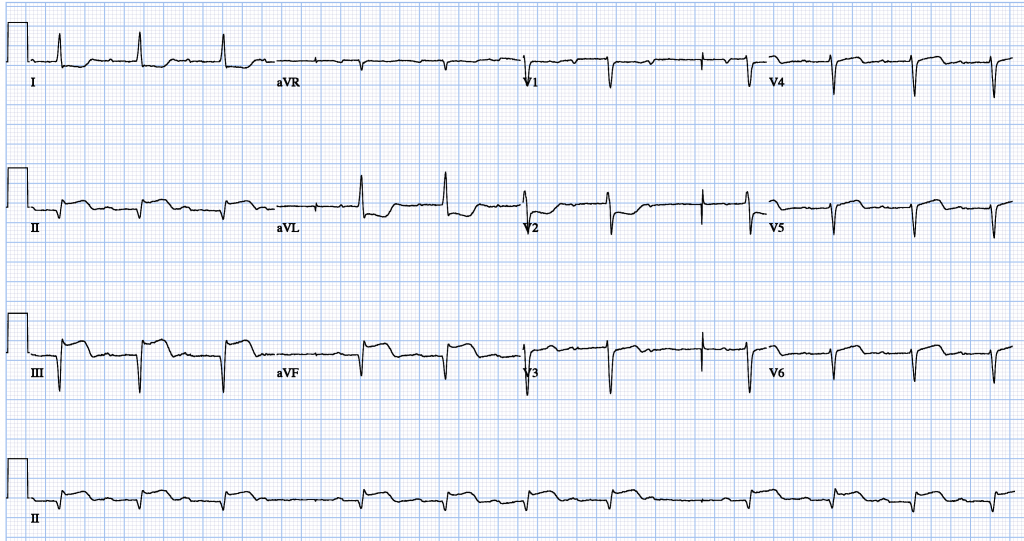
EKG Findings in RV Infarct
- Indirect evidence of RV infarction:
- ST elevation in lead III > Lead II (Suggests RCA occlusion)
- ST elevation in V1 (Remember: V1 is closest to the RV on a typical 12-lead EKG).
- ST elevation in V1 but ST depression in V2 (Suggestive of RV infarct).
- Direct evidence of RV infarction:
- ST elevation in V4R (V4 Reverse) (15-lead EKG is required). V4R puts a lead directly over the RV.
- Remember only 0.5mm of ST elevation is required to be considered positive in V4R.
Cath
- RCA lesions are most common reason for RV infarct as the RV branch is supplied by the RCA.
- Usually very proximal lesions.
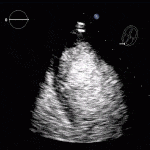
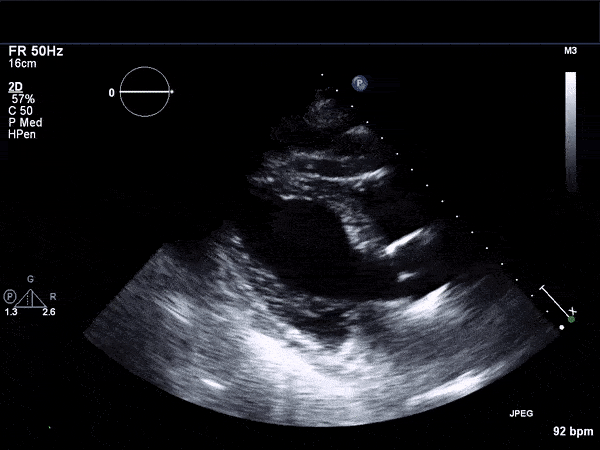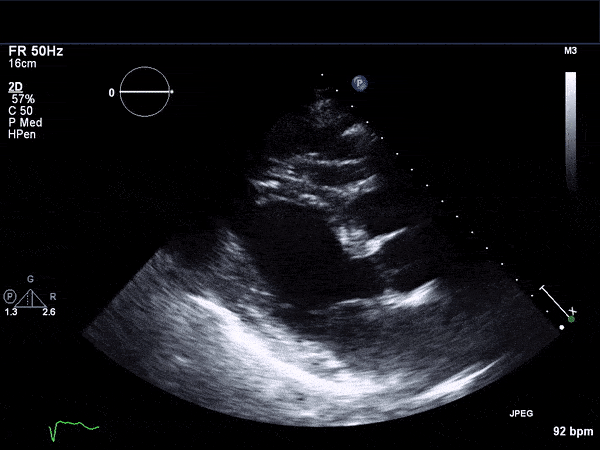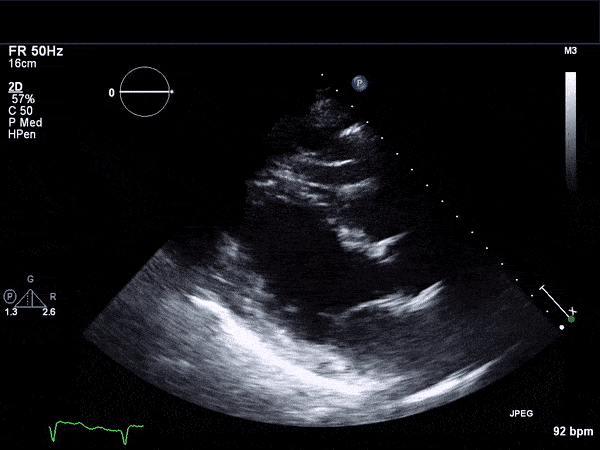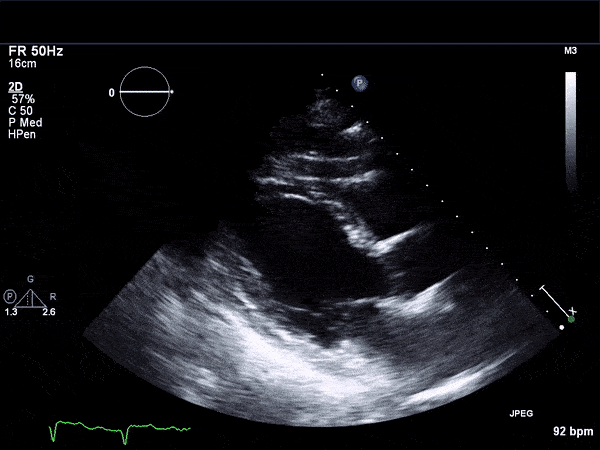
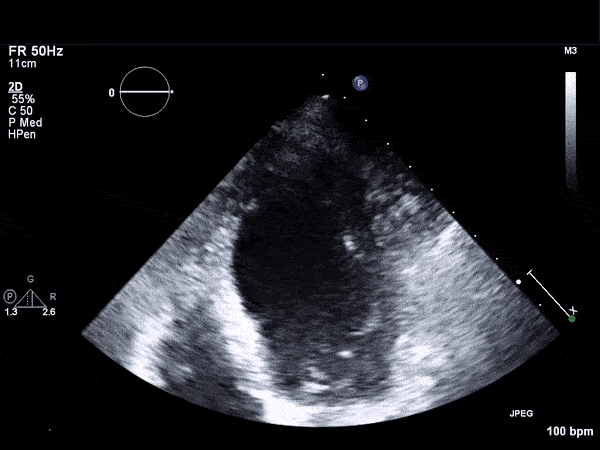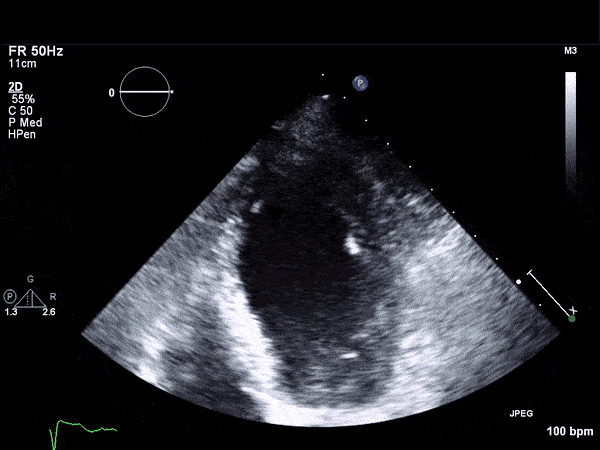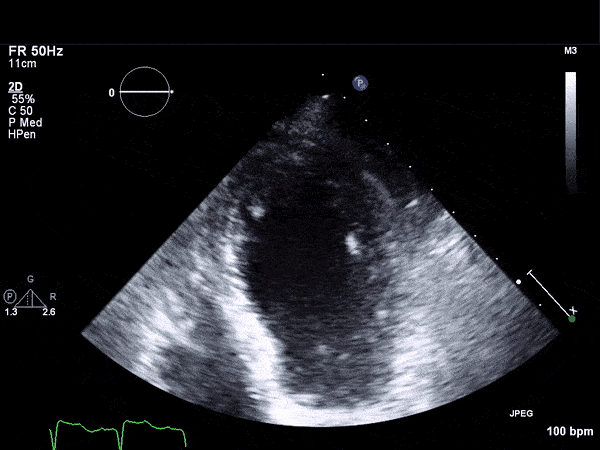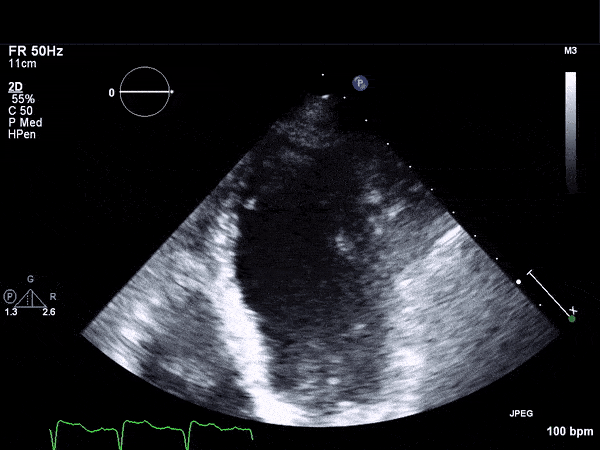
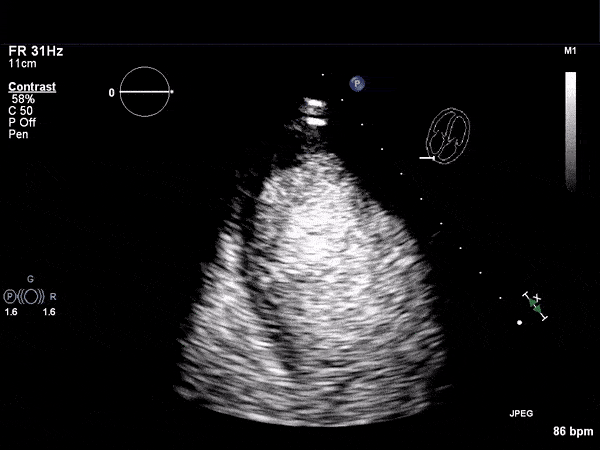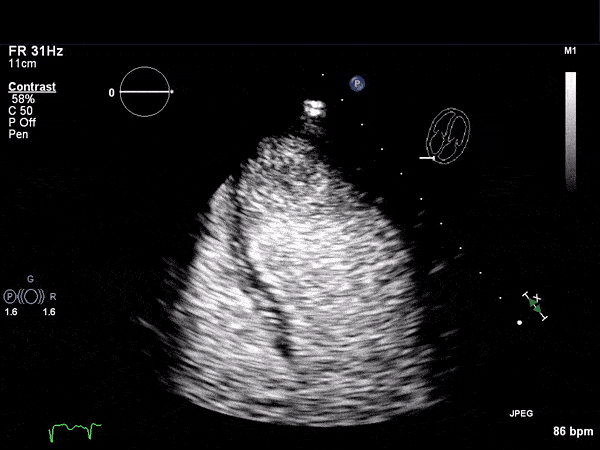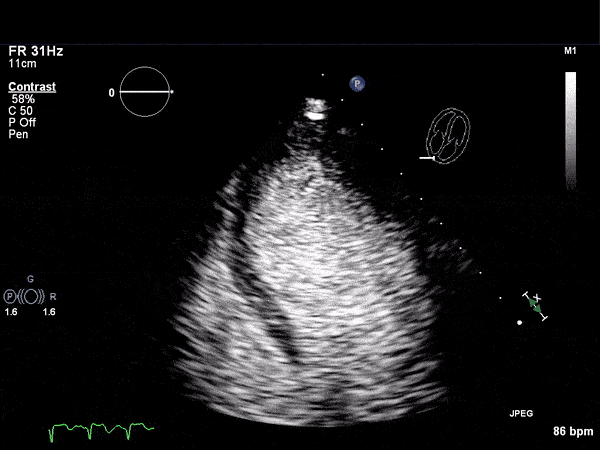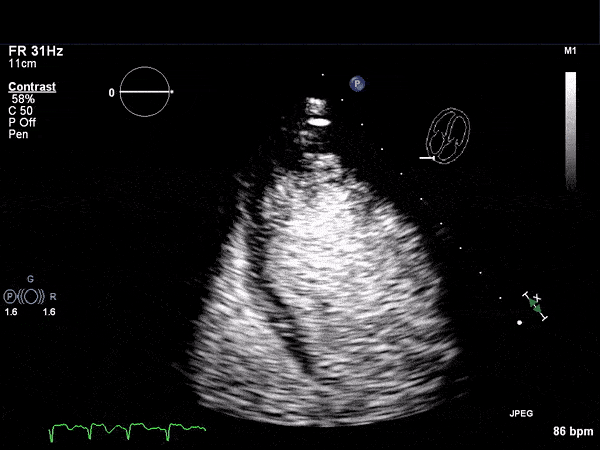
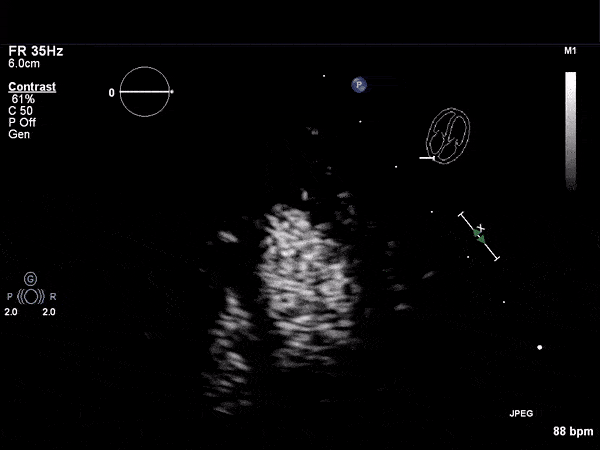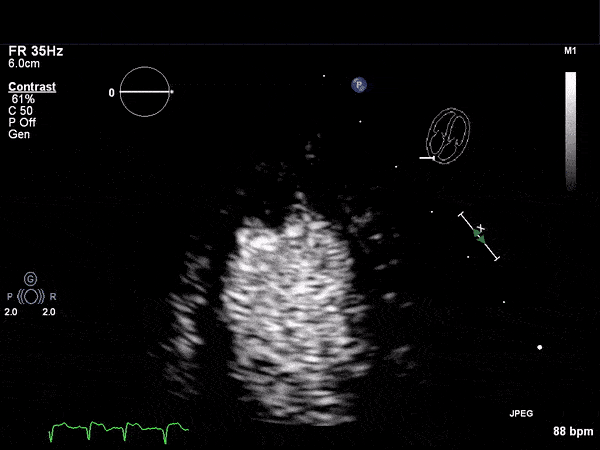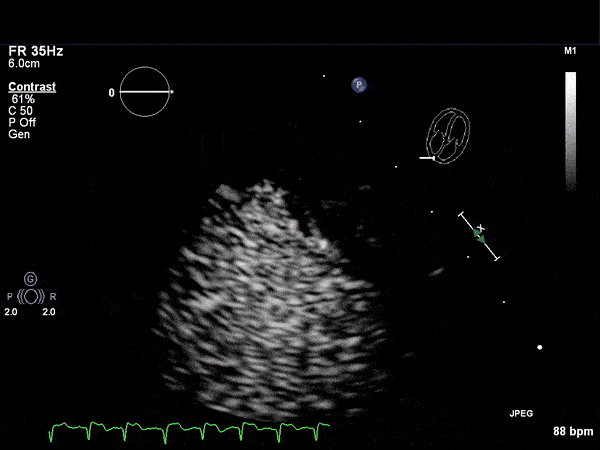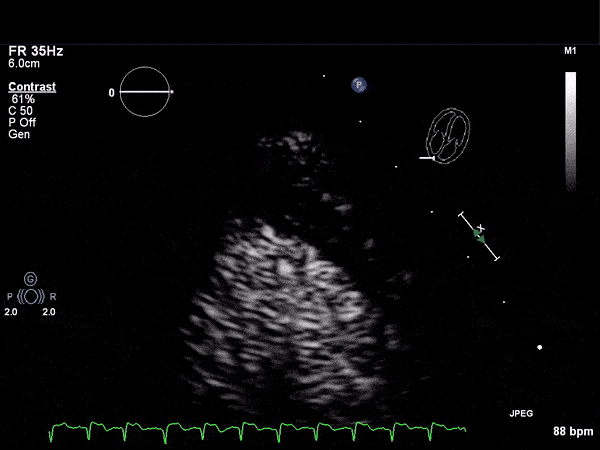
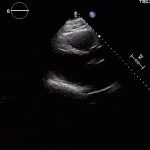
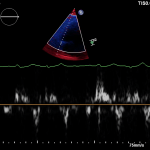
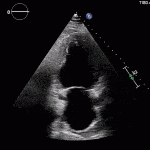
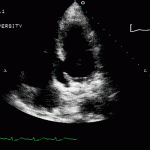
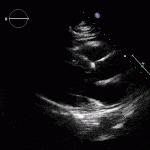
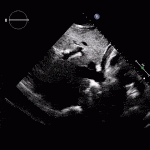
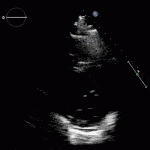
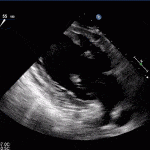
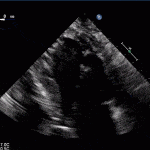
Echocardiogram
- Can be useful in assessing the severity of an RV infarct
- Visual qualitative assessment
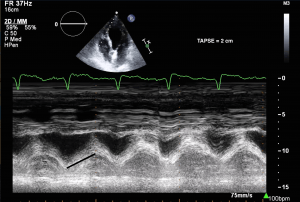
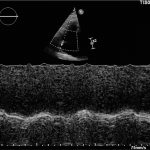
- TAPSE (Tricuspid Annular Plane Systolic Excursion) is a validated tool to assess RV function
- Involves am M-mode at the base of the RV to assess tricuspid annular motion
- Normal ≥ 1.7 cm
- TAPSE has been validated as a predictor of heart failure and survival
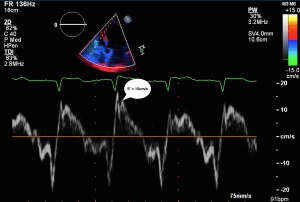
- Tissue Doppler
- Systolic excursion (S’) of < 9.5 cm/sec is suggestive of RV dysfunction
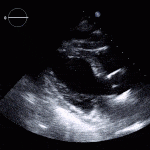
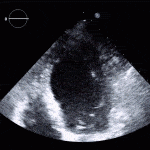
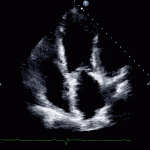
RV Infarct on ECHO
Management
- Revascularization
- Even after revascularization, the right ventricle may take some time to recover.
- Hypotension Management
- The main hemodynamic problem resulting from an RV infarction is reduced LV preload, which causes reduced cardiac output/shock and hypotension.
- Avoid therapies that further reduce preload (nitrates, narcotics, propofol, etc.)
- Use therapies to improve preload (IV fluids, vasopressors)
- Dobutamine
- Improves RV stroke volume, and improves RV afterload by reducing pulmonary vascular resistance
- Ideally used after revascularization, as inotropes can cause complications during acute MI (arrhythmias, increase in infarct size, etc.)
Heart Block
Inferior infarct-related heart block:
- Usually RCA, rarely can be circumflex
- Heart block associated with high-vagal tone Bezold-Jarisch reflex or rarely from AV node ischemia
- Often transient and can be treated with atropine
- Pacemaker should generally be avoided due to risk of RV perforation (can be placed in extreme circumstances)
Anterior infarct-related heart block:
- Usually related to extensive infarct and damage to the conduction system in the septum
- Complete heart block often preceded by RBBB + LAFB
- Temporary/permanent pacemaker more likely to be required; usually see more distal conduction disease like Mobitz 2, third degree heart block, or alternating bundle branch blocks
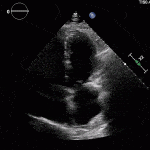
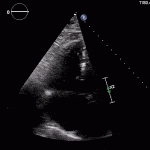
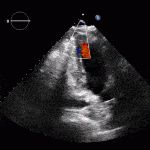
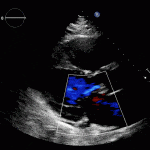
Ventricular Septal Defect
- A very uncommon complication of acute MI.
- Typically seen in late-presenting patients who have not been revascularized. They are often female, older patients, and have no history of MI/Angina prior to presentation (less collaterals).
- Often have poor prognosis.
Types
- Anterior Infarct often located at the apical ventricular septal.
- Inferior Infarct often located at the inferobasal ventricular septum. Usually a worse prognosis.
Clinical Features
- Prominent pan-systolic murmur at left lower sternal border often associated with a thrill.
- Signs of right sided heart failure, pulmonary edema or shock.
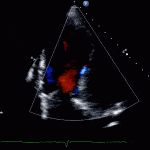
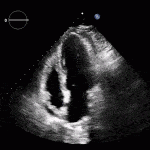
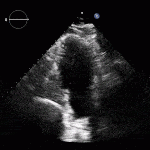
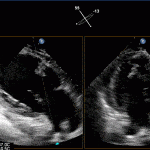
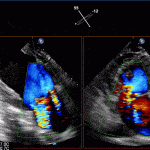
- Echo helps confirm evidence of a VSD with left to right shunt as well as complications such as RV failure.
Management
- Hemodynamic support with ionotropes, afterload reduction with nitroprusside and can consider IABP for mechanical support as bridge to surgery
- Surgical closure or percutaneous options
Myocardial Rupture
- Uncommon complication but very high mortality.
- The risk factors are similar to VSDs: patients are often older, late-presenting, female, first presentation MI (lack of collaterals)
- Patients can have sudden PEA arrest from cardiac tamponade
- Myocardial rupture is typically preceded by pain similar to the patient’s initial presentation
- Hypertension, excessive straining, coughing, or vomiting may be triggers
Management
- Cardiac Arrest → Standard ACLS plus pericardiocentesis
- Hemodynamic compromise → Fluids, dobutamine/pressors, pericardiocentesis
- Traditionally, pericardiocentesis was discouraged in patients with myocardial rupture due to concern of worsening hemorrhage into the pericardial space. However, pericardiocentesis (with removal of only 10-50 mL) is still sometimes performed in patients with extreme hemodynamic instability to improve hemodynamics until definitive surgical management is delivered.
- Surgical consultation is a must. Teflon or pericardial patch can be performed on the epicardium to stabilize the rupture.
Papillary Muscle Rupture
- Presentation:
- Acute severe pulmonary edema
- Hypotension
- Shock
- Murmur: may not be very loud because of rapid equalization of pressures across the mitral valve, but patients will have severe MR
- Posteromedial papillary muscle
- Most common
- Single blood supply PDA
- Anterolateral papillary muscle
- Dual blood supply from the LAD and circumflex artery
- Physical Exam / POC-US:
- Patients often have a hyperdynamic LV because severe MR reduces LV afterload, by allowing blood to pass to a low-pressure left atrium. This paradoxically improves LV systolic function, and it often becomes hyperdynamic (EF > 70%).
- Patients with acute MR develop acute severe pulmonary edema (“Flash pulmonary edema”) – they are acutely dyspneic requiring high O2.
- Physical exam will reveal acute congestion (pulmonary edema), and poor forward flow (cyanosis, pallor, confusion, lethargy, poor capillary refill).
- S3 “gallop” heart sound is very commonly auscultated, and is a classic finding.
- A murmur is very uncommon in severe MR because regurgitant flow is laminar across a wide-open mitral valve. Murmurs are produced by turbulent flow.
- Treatment
- Acute Stabilization:
- Afterload reduction (if blood pressure allows)
- Reducing afterload facilitates forward flow into aorta, and reduces regurgitation (often target MAP 60-65mmHg). However, the patient is often in shock and hypotensive, which precludes the use of vasodilators for afterload reduction. Afterload reduction to reduce MR must be balanced against severe hypotension, which reduces organ/coronary perfusion.
- Inotropes
- May need to combine with vasopressors to avoid severe hypotension.
- Inotropes may temporize the situation, but are unlikely to independently rescue a patient with acute MR.
- IABP
- Excellent solution to reduce afterload, and facilitate forward flow.
- IABP insertion is the best step to temporize a patient with acute severe MR.
- Afterload reduction (if blood pressure allows)
- URGENT surgical consult
- Patients with acute severe ischemic MR will not survive without EARLY surgical intervention.
- Cardiac surgery must be consulted WITHOUT DELAY.
- Acute Stabilization:
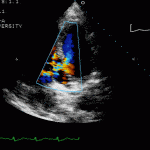
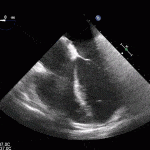
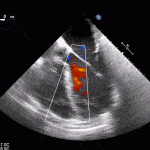
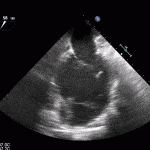
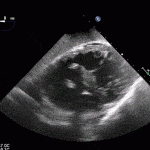
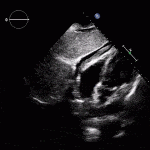
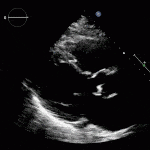
Transthoractic Echo
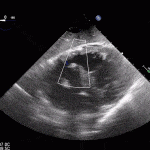
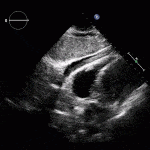
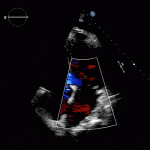
Transesophageal Echo
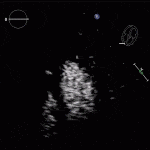
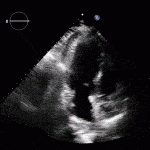
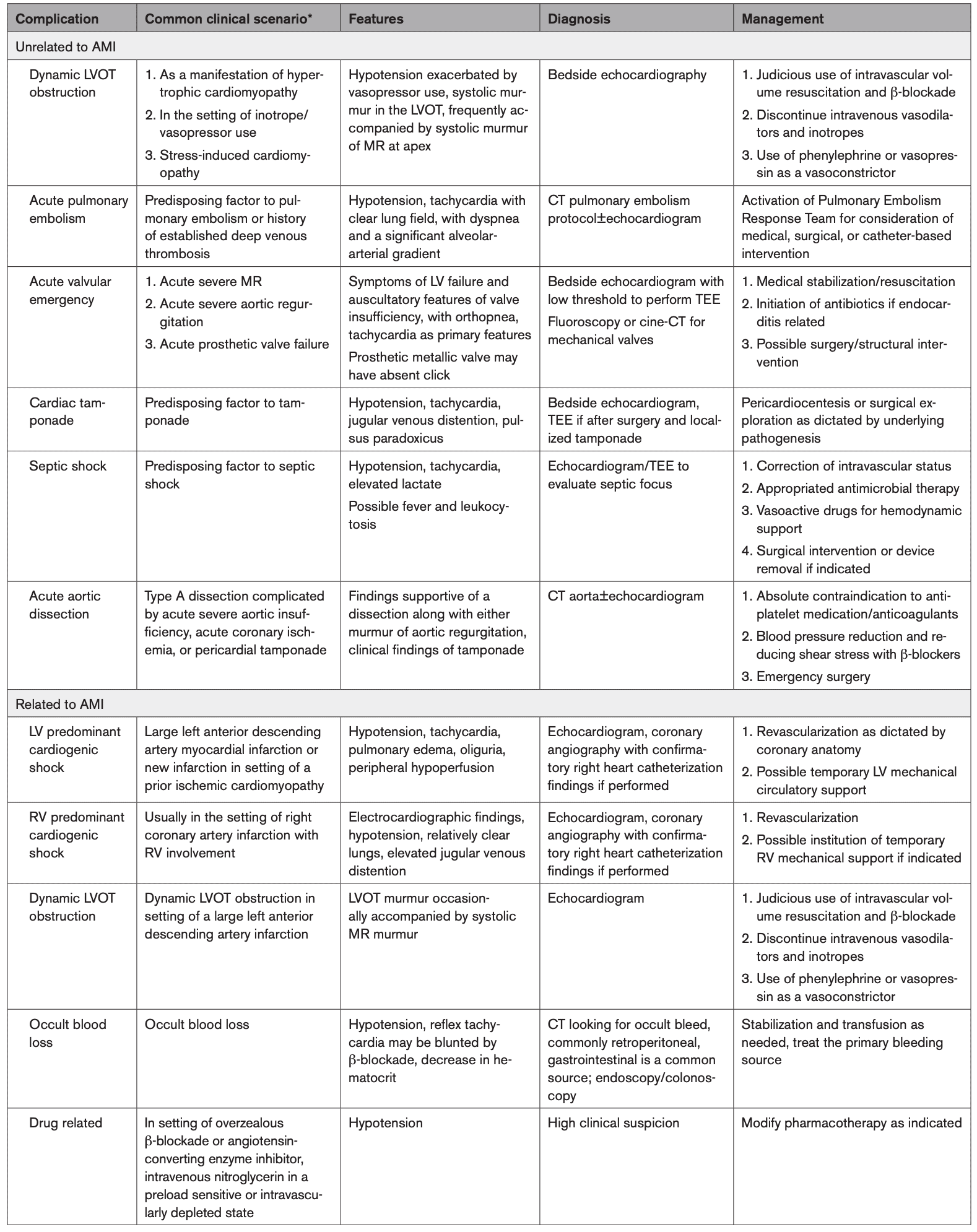
Differential
References
Figueras J, Cortadellas J, Soler-Soler J Left ventricular free wall rupture: clinical presentation and management Heart 2000;83:499-504.
- Delewi R, Zijlstra F, Piek JJ Left ventricular thrombus formation after acute myocardial infarction Heart 2012;98:1743-1749.
- Risk Factors, Angiographic Patterns, and Outcomes in Patients With Ventricular Septal Defect Complicating Acute Myocardial Infarction Brian S. Crenshaw et al.
Contributors
- Primary Authors:
- Dr. Daniel Durocher (MD, FRCPC)
- Dr. Pavel Antiperovitch (MD, FRCPC)
- Dr. Atul Jaidka (MD, FRCPC)
- Reviewers:
- Perri Deacon (medical student)