The Hemodynamic Problem
- Identifying and managing patients with CS requires an understanding of hemodynamics.
- Every patient must be carefully assessed – There is no one-size-fits-all approach with regard to treatment.
- Patients presenting with CS clinically behave very differently than other forms of shock, and are treated differently.
- To review:
| Shock | Preload | Cardiac output | BP | Treatment |
|---|---|---|---|---|
| Vasodilatory (i.e. sepsis) | Reduced | Reduced | Low |
|
| Hypovolemic | Reduced | Reduced | Low |
|
| Cardiogenic | HIGH | Reduced | Variable |
|
Approach to Management
The general approach to managing patients with CS involves concurrently addressing the following areas:
- Identify & Correct Reversible Causes
- Optimize Hemodynamics
- Medical Therapy for Cardiac Output
- Advanced Therapies / Palliation
1. Correct Underlying Cause
Patient’s survival depends on identification and correction of potential causes of CS. Delaying the diagnosis carries significant risks to the patient.
- Pulmonary Embolism must be ruled out (can mimic cardiogenic shock)
- Acute Myocardial Infarction
- Patients presenting with CS and STEMI must be revascularized as soon as possible.
- See “Revascularization” Section
- Acute Myocarditis
- Consider empiric immunosupression
- Acute Valve Dysfunction
- Urgent cardiac surgery consultation.
- Pericardial Effusion
- Perform pericardiocentesis as soon as possible.
- Arrhythmia / Bradycardia
- Electrical cardioversion
- Urgent transvenous pacing.
2. Optimize hemodynamics
Preload Optimization
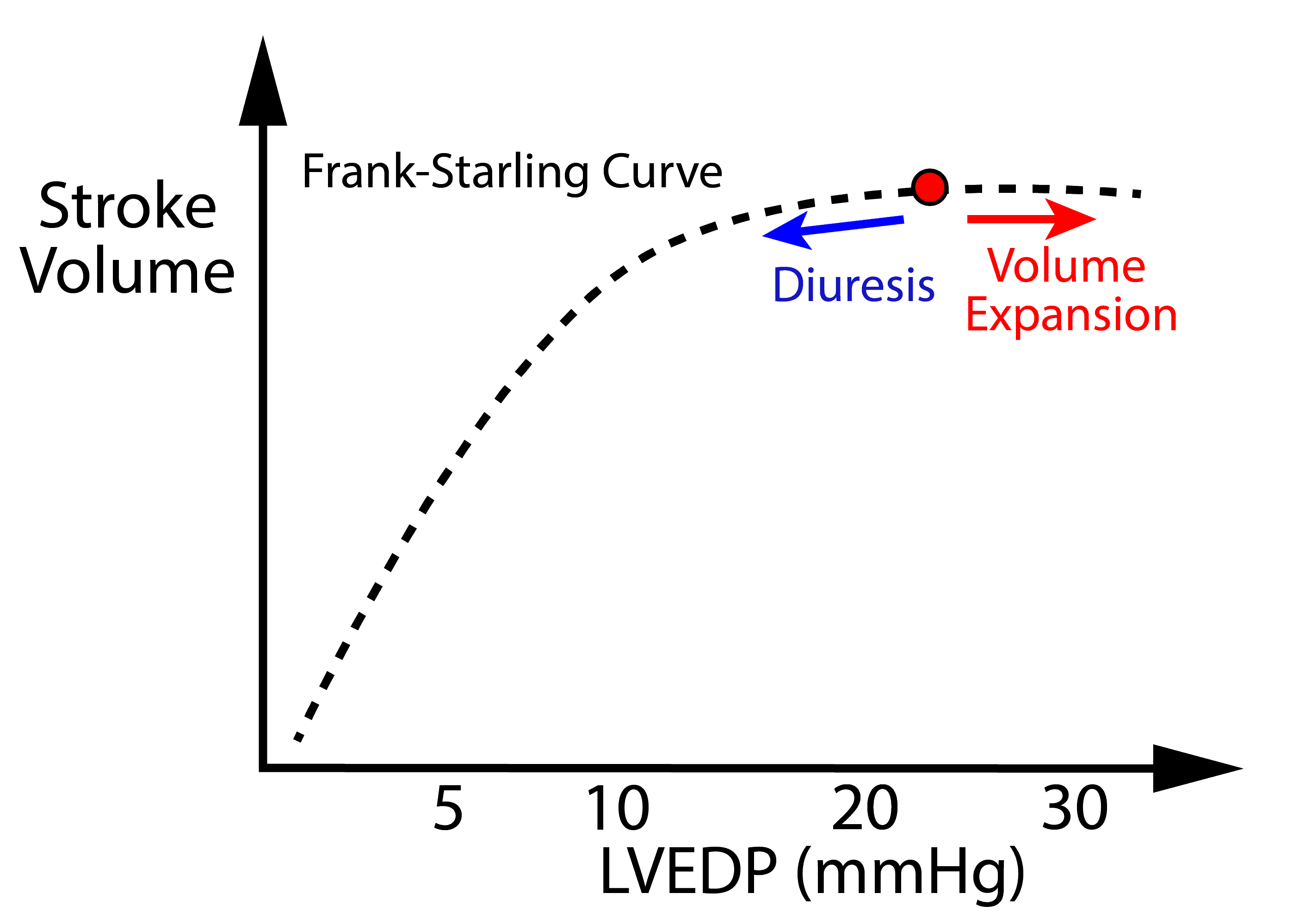
- The Preload Dilemma
- On one hand preload improves ventricular filling resulting in increased stroke volume – being mindful of diminishing returns based on Frank-Starling Law (see diagram).
- On the other hand, excess preload can cause hypoxemia due to pulmonary edema and also result in high venous pressures that can reduces tissue perfusion pressure.
- A careful balance of preload must be achieved.
- The vast majority of patients with CS have very high filling pressures (i.e. LVEDP of 25-40 mmHg) – the “cold & wet” phenotype. Further volume expansion with IV fluids is unlikely to improve stroke volume, and will result in:
- Worsening of hypoxemia due to pulmonary edema
- Reduction in tissue perfusion pressure by raising CVP.
- Small boluses of fluid can be considered for rare patients with CS who are “cold and dry” – especially if they do not require supplemental oxygen.
- Keep in mind that fluid is easy to give, but very challenging to remove.
- Decongestion can be achieved by:
- Diuresis
- IV furosemide (5-20 mg/h) – infusion preferred by many Cardiologists
- IV bumetanide – if patients are furosemide resistant.
- Oral metolazone (2.5-5mg) – can be added to furosemide for synergistic effect.
- NOTE: diuretics are often not effective in CS due to reduced renal perfusion and strong fluid-retention mechanisms (i.e. RAAS). Ultrafiltration is often required.
- Ultrafiltration (dialysis)
- Allows controlled removal of fluid.
- Must be done gently to minimize large changes in preload causing hypotension.
- Diuresis

Afterload Reduction
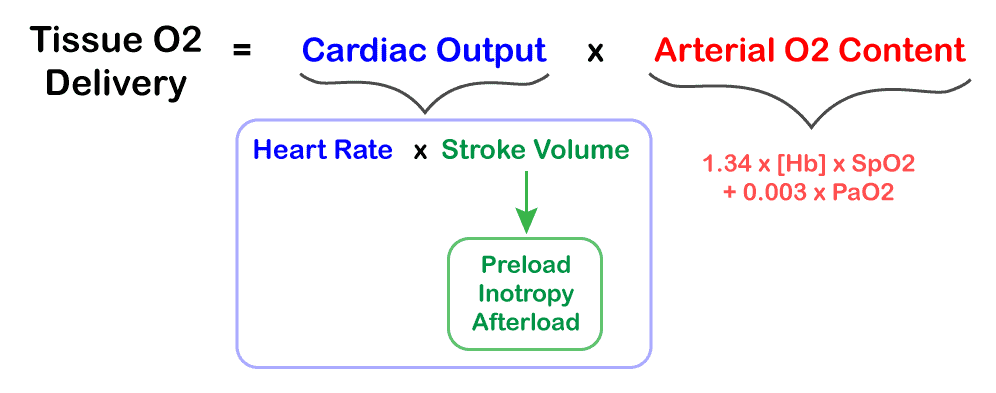
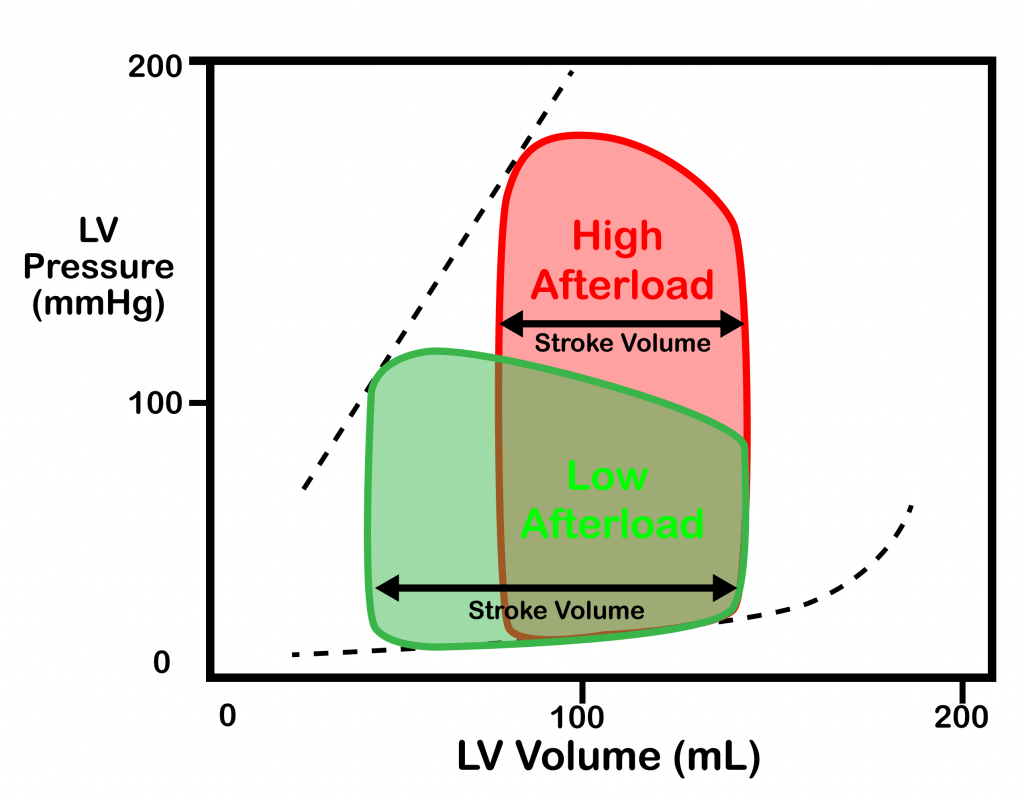
- Reduction of afterload has the following beneficial effects:
- Improves stroke volume / cardiac output (see diagram for ventricular hemodynamics).
- Reduces cardiac work
- Reduces myocardial oxygen requirements
- Improves myocardial perfusion (by reducing wall tension)
- However, a very low MAP can result in poor tissue/coronary perfusion pressure, and can contribute to ischemia. A careful balance must be achieved.
- A lower MAP should be targeted in patients with CS compared to patients with other types of shock. The exact number is not well established and must be tailored to the patient. Many clinicians use ~ 60-65mmHg as a general target.
- NOTE: Many patients with CS are hypotensive, and afterload reduction is not possible. If hypotension is significant, a vasopressor must be used to raise perfusion pressure.
Control of MAP can be achieved by:
- Vasopressors to increase MAP
- Norepinephrine is most preferred due to some inotropic effects and limited evidence.
- Other options include vasopressin and epinephrine. Many clinicians avoid phenylephrine in CS due to pure vasoconstrictive effect without any inotropic effects.
- SOAP II trial — norepinephrine as first line agent in patients with shock was associated with less arrhythmia vs. dopamine [in the CS subgroup]
- Vasodilators to reduce MAP
- Prefer short acting vasodilators that are easy to titrate.
- Many Cardiologists prefer nitroprusside due to its short half-life.
- NOTE: An arterial line is mandatory for careful titration.
- NOTE: Invasive hemodynamic monitoring with a Swan-Ganz catheter can allow objective measurements + titration of filling pressures. Although routine use of Swan-Ganz catheters in septic shock and ARDS did not improve outcomes and had potential of harm (ARDS JAMA 2003), patients with cardiogenic shock are generally excluded from these studies.
Special Case: RV Failure
- Patients with isolated RV failure have normal or reduced left sided pressures (LA pressure, LVEDP, pulmonary capillary), and commonly present with significant hypotension.
- Due to low left-sided filling pressures, increasing preload with intravascular volume expansion can further increase overall cardiac output by improving LV filling. This carries a much lower risk of pulmonary edema.
- Note: pulmonary edema is still possible, and can eventually develop if the LV filling pressure rises or oncotic pressures drops. Patients must be carefully monitored for worsening hypoxemia when treated with fluids.
- CAVEAT: Patients with chronic RV failure already have adapted to an extremely very high preload. Delivering extra fluid to those patients may not result in significant gain in cardiac output, and may worsen tissue perfusion pressure by increasing CVP.
- Do not give fluid to patients who are chronically volume overloaded without expert advice.
Special Case: Pericardial Disease
- When there is increased external pressure on the heart, chambers with the lowest cavity pressures are affected first –> RA and RV.
- Hemodynamic instability (mainly hypotension) from pericardial tamponade and constriction is usually caused by reduced oRV filling. These patients respond to fluids without a concern for pulmonary edema just like other patients with isolated RV failure patients. However, their oxygen requirements must be carefully monitored.
3. Increase Cardiac Output (Medical)
- Inotropes can be started to improve cardiac output.
- Risks: Inducing arrhythmia, increasing myocardial oxygen requirements.
- Pure inotropes readily available in Canada include Milrinone and Dobutamine, and cause vasodilation in addition to inotropy (aka “inonodilator”). They help reduce afterload.
- If afterload reduction is not desired, these inotropes can be paired with vasoconstrictors such as norepinephrine.
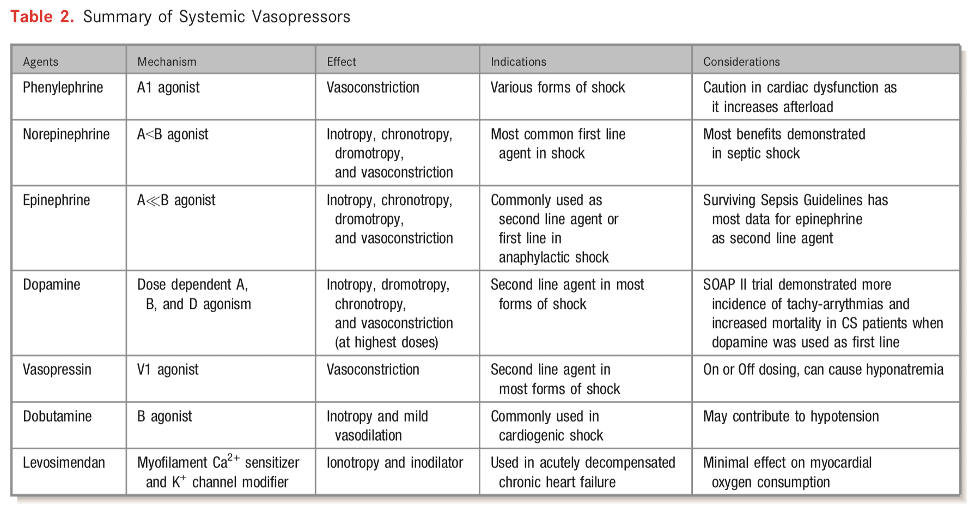
- Pleaser review the “Mechanism of Action of Common Vasoactive Agents” chart.
Property Dobutamine Milrinone Onset/Half-life - FAST Onset (1-10min)
- Short Halflife (2 min)
- Slower Onset
- Longer Halflife (3 hrs)
- Halflife up to 20h if on dialysyis or poor renal function
Hypotension (SVR Reduction) - Mild vasodilator though B-2 receptors.
- Peripheral and pulmonary vasodilator
- If vasodilation is not desired, often paired with vasopressor
Renal Failure - Not renally cleared
- Renally cleared, accumulates in renal dysfunction
- Half-life can be up to 20hrs in dialysis/severe renal dysfunction (needs dose-adjustment)
- Poorly dialyzable (depends on type of dialysis – i.e. CVVH)
Beta-blocker Use - Chronic beta-blocker use down-regulates Beta-receptors. Dobutamine may be theoretically less potent (need to use higher dose)
- Downstream effects of beta-receptor (not affected by beta-blockers)
4. Advanced Therapies / Palliation
Mechanical support
- Paucity of high-quality evidence to support the routine use of MCS as therapeutic adjunct
- AHA recommendation — “patients with persistent CS, with or without end-organ hypoperfusion, should be evaluated for MCS candidacy by multidisciplinary team with expertise in the selection, implantation, and management of MCS devices”
- MCS for:
- Bridge to recovery
- Bridge to bridge
- Bridge to transplant
- Bridge to decision
- INTERMACS registry reveals decline in use of durable MCS for INTERMACS profile 1 and 2 patients due to high mortality rates despite MCS implantation (38% 30d mortality)
- Consequently, recommendations support use of temporary MCS for INTERMACS profile 1 and 2
- Select temporary MCS options
- IABP — CO 0.5-1L/min
- Most widely used temporary MCS device
- 7-8Fr sheath
- Impella 2.5, Impella CP — 2.5-4L/min
- 11Fr sheath
- Impella 5 — 5L/min
- 22Fr sheath requiring surgical cutdown
- Non-durable VADS
- Tandem Heart
- Centrimag
- VA ECMO
- Used to support both CV system and respiratory system
- AHA statement –> “VA-ECMO should be the preferred temporary MCS option when there is poor oxygenation that is not expected to rapidly improve with an alternative temporary MCS device or during resuscitation”
- Relative contraindications
- Age > 75
- Life expectancy < 1 year
- Severe PVD
- Advanced liver disease
- Contraindications to systemic anticoagulation
- Neurological injury / disability
- Complications
- Distal limb ischemia
- Thromboembolism
- Stroke
- Bleeding
- Hemolysis
- Infection
- AV insufficiency
- Increased LV afterload –> inadequate unloading of LV
- Usually requires –> IABP / Impella, atrial septostomy
- IABP — CO 0.5-1L/min
Durable MCS & transplantation
-
- Reviewed elsewhere
Reperfusion and Revascularization
- The mainstay evidence-based therapeutic intervention for patients with AMI and CS
- Fibrinolytic therapy
- Very few placebo controlled trials have enrolled patients with CS
- GUSTO-1 trial –> no mortality benefit in any fibrinolytic strategy in CS (approx. 3000 patients)
- Only considered in patients with STEMI + CS in whom invasive approach in timely fashion is not possible
- PCI strategy
- SHOCK trial –> benefit to PCI in CS
- ? Culprit vs. Complete revascularization in CS
- CULPRIT-SHOCK –> benefit for culprit-only PCI compared to complete revasc.
- Discordant results in DANAMI-3, PRAMI, CvLPRIT
- KAMIR-NIH –> benefit for complete revasc. compared to culprit-only
- OVERALL –> discordant results; need for more data
- Evaluate on case-by-case basis
- CULPRIT-SHOCK –> benefit for culprit-only PCI compared to complete revasc.
- Early invasive vs. delayed strategy in CS
- SMASH –> no significant reduction in mortality in early invasive strategy (only 55 patients)
- SHOCK –> no difference in 30d all-cause mortality; however, lower mortality at 6 and 12 months in early invasive arm (302 patients)
- OVERALL –> AHA recommends early invasive strategy for all suitable patients with suspected ACS-associated CS
- Multiple classifications exist:
- Two uncommon but hemodynamically distinct entities:
- Normotensive CS
- 2% of the SHOCK trial
- Peripheral hypoperfusion, despite SBP >90
- Comparable CI, PCWP, LVEF vs. other patients, but higher SVR
- Highlights importance of “relative” hypotension***
- Right ventricular CS
- 3% of the SHOCK trial
- Severity of shock may depend of degree of both RV and LV ischemia
- Characterized by –> higher CVP, LVEF; lower PASP
- Similar CI, PCWP
- Classic hemodynamic definition of RV infarction
- CVP / PCWP >/= 0.8
- Normotensive CS
Further Reading
- Contemporary Management of Cardiogenic Shock: A Scientific Statement From the American Heart Association. Sean Van Diepen et. al. 2017. (html)
- Cardiogenic Shock. American Heart Association. Valdahpour et al. 2019. (html)
- SCAI Clinical Expert Consensus Statement on the Classification of Cardiogenic Shock. Catheterization and Cardiovascular Interventions. David Baren et al. 2019. (html)
- Epinephrine Versus Norepinephrine for Cardiogenic Shock After Acute Myocardial Infarction. Journal of American College of Cardiology. Bruno Levy et al. 2018. (html)